 The background color scheme of cells categorizes the 118 elements into four groups: (1) white indicates the element has no standard atomic weight, (2) blue indicates the element has only one isotope that is used to determine its standard atomic weight, which is given as a single value with an uncertainty, (3) yellow indicates the element has two or more isotopes that are used to determine its standard atomic weight, which is given as a single value with an uncertainty, and (4) pink indicates the element has a well-documented variation in its atomic weight, and the standard atomic weight is expressed as an interval.Īn element-by-element review accompanies the IPTEI and includes a chart of all known stable and radioactive isotopes for each element. Color-coded pie charts in each element cell display the stable isotopes and the relatively long-lived radioactive isotopes having characteristic terrestrial isotopic compositions that determine the standard atomic weight of each element. The IPTEI is intended to hang on the walls of chemistry laboratories and classrooms.Įach cell of the IPTEI provides the chemical name, symbol, atomic number, and standard atomic weight of an element. The IPTEI is modeled on the familiar Periodic Table of the Chemical Elements. eTOC ‘Chemistry International’ – Jan-Mar 2024 Ībstract: The IUPAC Periodic Table of the Elements and Isotopes (IPTEI) was created to familiarize students, teachers, and non-professionals with the existence and importance of isotopes of the chemical elements.The 2024 IUPAC-Richter Award Goes to Craig M. /PeriodicTablenamescolor-58b5d91e3df78cdcd8d03326.jpg)
IUPAC Standards Online – FREE ACCESS IN 2024.2024 Franzosini Prize and Balarew Award – Call for Nominations.Inorganic Chemistry Division – Feb 2024 Newsletter.eTOC Alert ‘Pure and Applied Chemistry’ – January 2024.Note that each of these entries corresponds to one of the twelve periods respectively. Group six (R H superscript two, R 0 superscript three) contains the following information: (no entry for period 1), O = 16, S = 32, C r = 52, S o = 78, M o = 96, T o = 125, -, -, W = 184, -, U = 240. Group five (R H superscript two, R superscript two 0 superscript five) contains the following information: (no entry for period 1), N = 14, P = 31, V = 51, A s = 75, N b = 94, S b = 122, -, -, T a = 182, B l = 208,. Group four (RH superscript four, R0 superscript eight) contains the following information: (no entry for period 1), C = 12, B i = 28, T i = 48, - = 72, Z r = 90, S n = 118, ? C o = 140, ? L a = 180, P b = 207, T h = 231. Group three (-, R superscript one 0 superscript nine) contains the information: (no entry for period 1), B = 11, A l = 27, 8. Note the ach of these entries corresponds to one of the twelve periods respectively. The second group (-, R 0) contains the following information: (not entry for period 1) B o = 9, 4, M g = 24, C a = 40, Z n = 65, S r = 87, C d = 112, B a = 187, -, -, H g = 200,. 
Figure B shows the first periodic table developed by Mendeleev, which had eight groups and twelve periods. 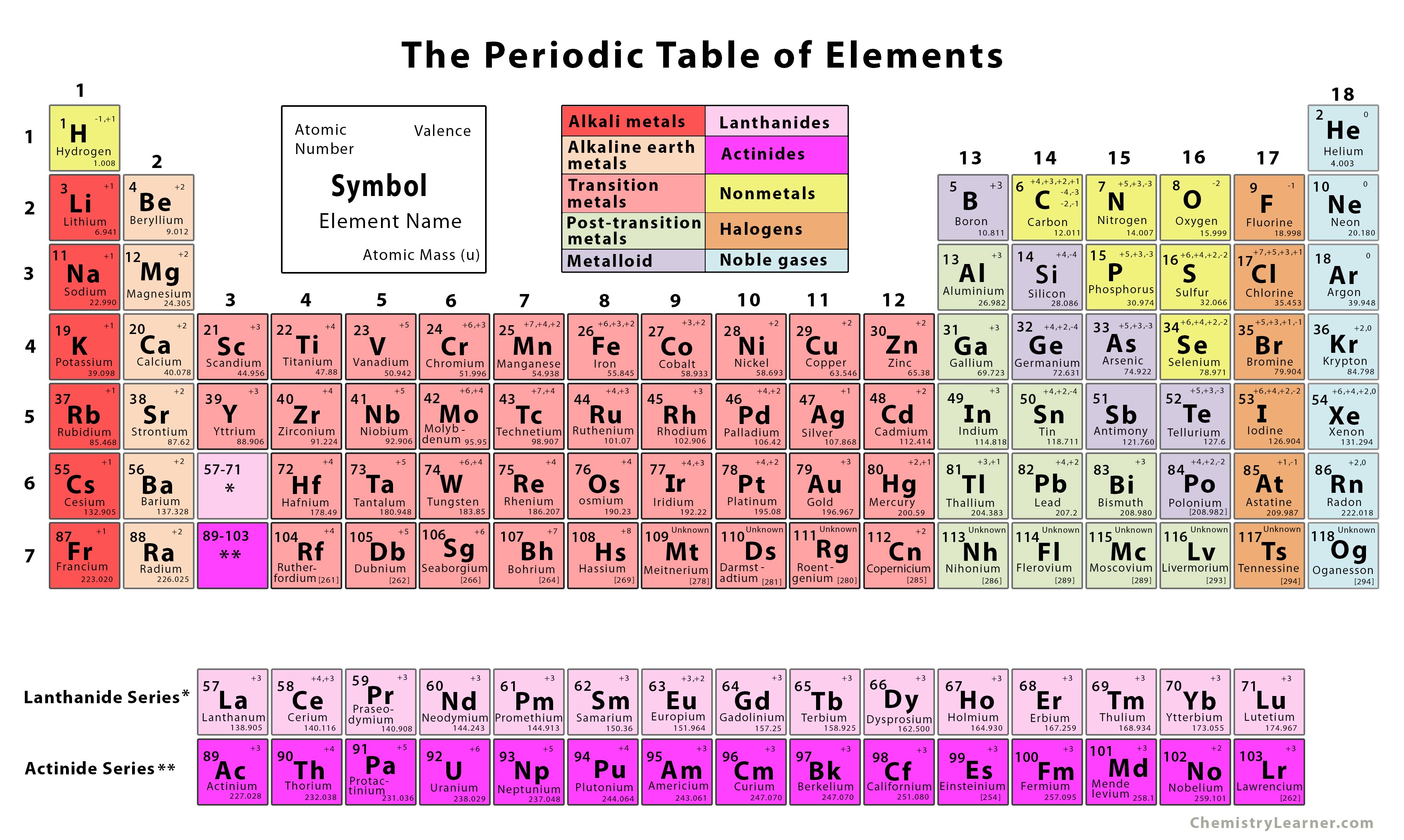
(credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons) Figure A shows a photograph of Dimitri Mendeleev. \): (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
